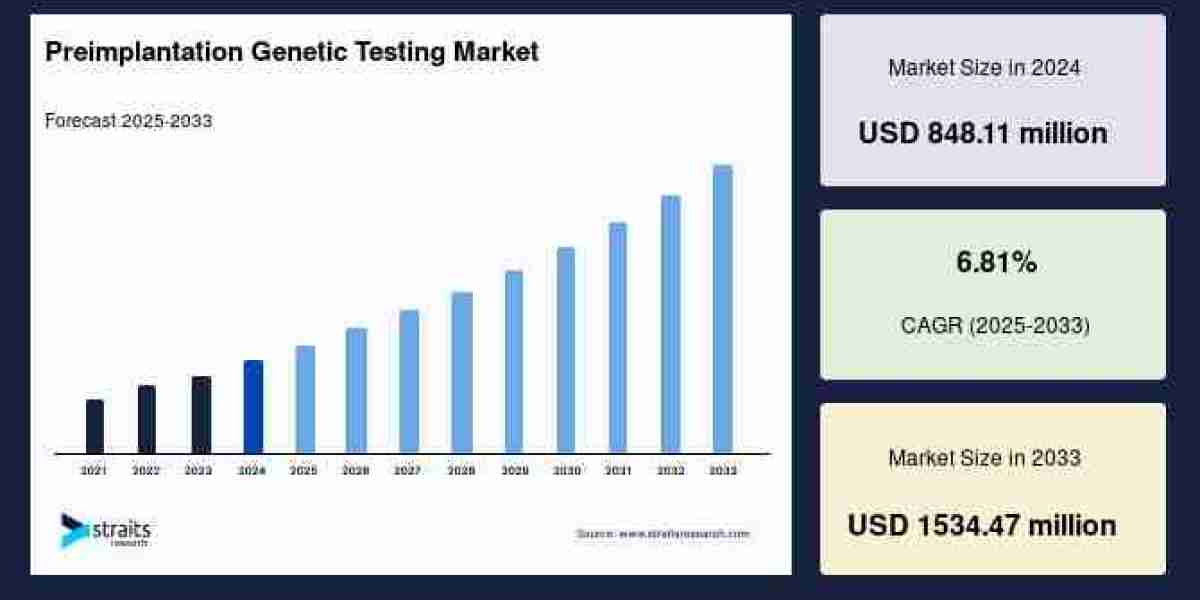
The medical polyoxymethylene (POM) market has emerged as a niche yet increasingly critical segment within the broader healthcare materials industry. Polyoxymethylene, also known as acetal or polyformaldehyde, is an engineering thermoplastic distinguished by its exceptional mechanical strength, rigidity, dimensional stability, low friction, and chemical resistance. These properties make POM particularly well‑suited for high‑precision and high‑performance medical applications where durability and sterilization compatibility are essential. Over recent years, demand for medical‑grade POM has been propelled by rapid developments in medical devices, the aging global population, and evolving healthcare delivery models that emphasize advanced, reliable components.
According to industry estimates, the global medical polyoxymethylene market was valued at around USD 1.8 billion in 2024, with projections indicating it could expand to more than USD 3.2 billion by the mid‑2030s, growing at a compound annual growth rate of approximately 5 % to 7 % through the forecast period. Market size projections vary depending on methodology and segmentation, but consensus points to steady, sustained growth driven by a combination of technological adoption, regulatory compliance, and increasing requirements for precision medical components.
The intrinsic balance between performance and processability is central to POM’s appeal in medical settings. It exhibits excellent machinability and can be precision‑molded into intricate parts with tight tolerances, which is crucial for components in devices such as drug delivery systems, surgical instruments, and diagnostic equipment. Moreover, medical‑grade POM conforms to stringent safety and biocompatibility standards — including USP Class VI and FDA approvals — enabling its use in direct patient contact applications. Its resistance to sterilization methods such as gamma radiation, steam, and ethylene oxide further elevates its suitability for reusable and single‑use medical components.
One of the primary drivers of market growth is the rising demand for precision medical devices. Minimally invasive surgery has become a dominant trend in modern healthcare, offering patients reduced recovery times, minimized surgical trauma, and lower risk of infection. These procedures depend on lightweight, durable, and highly reliable materials to manufacture specialized instruments. Polyoxymethylene fits this niche due to its ability to maintain dimensional integrity under mechanical stress and repeated sterilization cycles. As a result, the adoption of POM in surgical tools for laparoscopic, endoscopic, and robotic procedures continues to increase.
The proliferation of chronic diseases globally — including diabetes, cardiovascular disorders, and respiratory conditions — also shapes demand. For example, devices such as insulin pens, inhalers, and other drug delivery mechanisms have become ubiquitous in therapeutic regimens, particularly for long‑term outpatient care. These devices rely on POM for their internal mechanisms due to its low friction coefficients, resistance to wear over repeated use, and capacity for reliable performance even at extremely small scales. The growing elderly population contributes to this trend, as older adults typically require more frequent medical interventions and benefit from the convenience and accuracy offered by advanced drug delivery technologies.
Simultaneously, the healthcare sector’s structural transformation towards home healthcare and point‑of‑care diagnostics fuels demand for components manufactured from medical POM. Home monitoring tools, wearable medical devices, and patient self‑care systems increasingly employ high‑performance plastics to achieve compact, comfortable, and reliable designs. The market for diagnostic cartridges, microfluidic cartridges, and other precision components suitable for rapid, onsite testing has expanded, particularly in emerging and middle‑income regions.
Regional dynamics illustrate a diverse picture of growth and opportunity. Asia‑Pacific has emerged as one of the fastest‑expanding markets, driven by rising healthcare infrastructure investment, expanding domestic manufacturing capabilities, and growing focus on export‑oriented medical device production. Countries such as China, India, Japan, and South Korea have actively promoted medical technology development and have responded to rising internal demand for advanced medical solutions. Increased healthcare spending, paired with policy initiatives that support local production, has stimulated adoption of medical‑grade polymers, including POM.
In North America and Europe, mature healthcare systems and well‑established regulatory frameworks have created stable demand for POM materials, especially among leading medical device OEMs (original equipment manufacturers) seeking reliable and compliant material sources. These regions are also significant because of the presence of stringent quality and safety standards, which necessitate sophisticated material documentation and traceability, reinforcing the competitive advantage of certified medical POM suppliers. The emphasis on reducing device failures, minimizing recalls, and ensuring consistent delivery performance further integrates POM selection early in design and development cycles of medical products.
Despite these positive growth drivers, the medical polyoxymethylene market is not without challenges. Specialized medical‑grade POM is inherently more expensive to produce than general‑purpose grades due to rigorous testing, certification requirements, and high raw material purity specifications. These costs can represent barriers in cost‑sensitive healthcare environments or markets where alternatives may suffice for certain non‑critical applications. Additionally, fluctuating raw material prices and manufacturing costs can impact profitability and supply chain stability.
Competition from other engineering plastics and materials such as polycarbonate, polyethylene, and metals continues to influence market trends. While POM offers a unique combination of properties, choices in material selection often come down to application‑specific requirements and cost constraints. In certain implantable applications or where ultra‑long‑term biostability is required, alternative materials may be more appropriate.
Material innovation remains a focal point for market participants seeking to differentiate their offerings and capture greater market share. Recent advancements have seen the development of specialized POM grades with enhanced biocompatibility, improved sterilization tolerance, and lower extractable and leachable profiles. Some newer formulations also incorporate antimicrobial additives or radiopaque fillers to broaden application capabilities. Innovations in additive manufacturing have expanded the scope for customized and patient‑specific medical components, although POM’s crystallinity and processing complexities present ongoing challenges for 3D printing technologies. Nonetheless, progress in compatible 3D printing filaments and selective manufacturing methods suggests future opportunities for design flexibility and rapid prototyping in medical device production.
Sustainability considerations in healthcare are also affecting market trajectories. Hospitals and healthcare providers increasingly prioritize materials that support efficient sterilization, reduced waste, and potential recyclability. While traditional medical POM is not widely recyclable in standard hospital waste streams, pilot programs and closed‑loop recycling initiatives are gaining attention. Bio‑based POM derived from renewable feedstocks is under research and development, offering potential reductions in carbon footprint and aligning with environmental, social, and governance (ESG) goals that many medical device manufacturers and procurement organizations are now adopting.
In terms of competitive landscape, the market for medical polyoxymethylene features several global players engaged in ongoing innovation, strategic partnerships, and supply agreements with medical device manufacturers. These collaborations often extend beyond bulk resin supply to include dedicated R&D support, regulatory compliance assistance, and material customization services tailored to precise device needs. Such vertical integration helps ensure consistent quality, faster validation timelines, and material readiness aligned with medical device regulatory submissions.
Looking ahead, the medical POM market is poised for continued expansion as healthcare systems evolve and the reliance on high‑performance materials grows. Demand for minimally invasive procedures, chronic disease management tools, home healthcare devices, and advanced diagnostics will sustain a strong growth trajectory. Technological advancements, regional healthcare investments, and ongoing material development will all contribute to shaping a resilient and dynamic market landscape.
In summary, medical polyoxymethylene holds a strategic position within the medical plastics industry due to its combination of performance, reliability, and versatility. As the global healthcare market continues its transformation toward more sophisticated and patient‑centric solutions, POM’s role in enabling precision, durability, and safe medical component performance will be increasingly recognized. While challenges persist in cost and manufacturing complexity, innovation and regulatory alignment will support the material’s continued adoption across a wide array of medical applications, reinforcing its significance in the medical materials market of the future.